CBC options at IDEXX Reference Laboratories provide diagnostic flexibility
By M. Alexis Seguin, DVM, MS, DACVIM
Accurate and comprehensive
All complete blood counts (CBCs) performed at IDEXX Reference Laboratories use the most advanced hematology technology available. Fluorescent laser flow cytometry methods and veterinaryspecific algorithms make it possible to provide automated CBC results with an extremely high level of accuracy.
IDEXX CBCs are also the most comprehensive. They are the only CBC to include both:
- Reticulocyte count, regardless of the presence of anemia. A reticulocyte count on anemic patients helps to classify the anemia as regenerative or nonregenerative. In nonanemic animals, a persistent reticulocytosis may indicate compensated occult blood loss or underlying hemolytic disease.
- Reticulocyte hemoglobin, a measure of the quality of the developing red blood cells in canines and felines. The reticulocyte hemoglobin measures iron availability in developing red blood cells. A decrease in reticulocyte hemoglobin can be seen with blood loss or inflammation in anemic and nonanemic patients.
Flexible, easy-to-understand options
IDEXX Reference Laboratories offers the following CBC ordering options to give you the diagnostic flexibility you need to meet the individual needs of your patients.
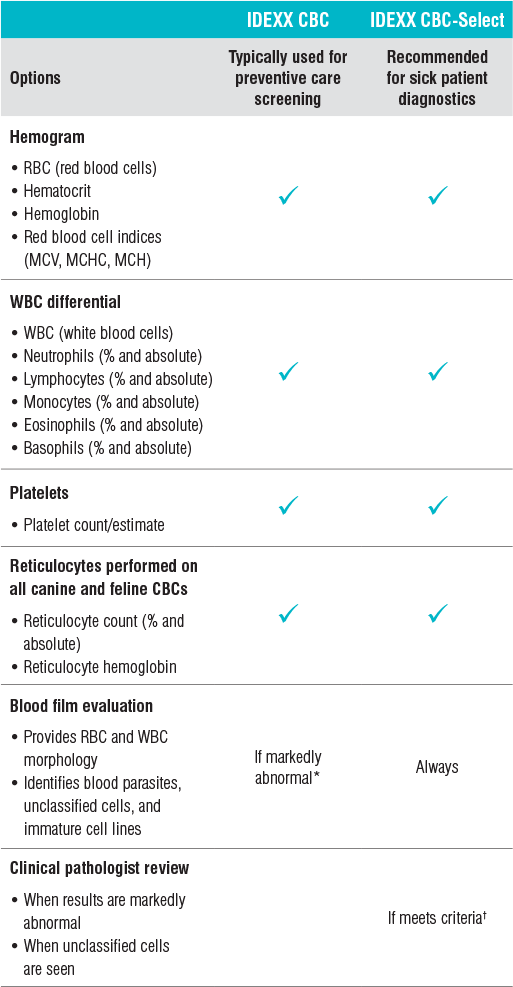
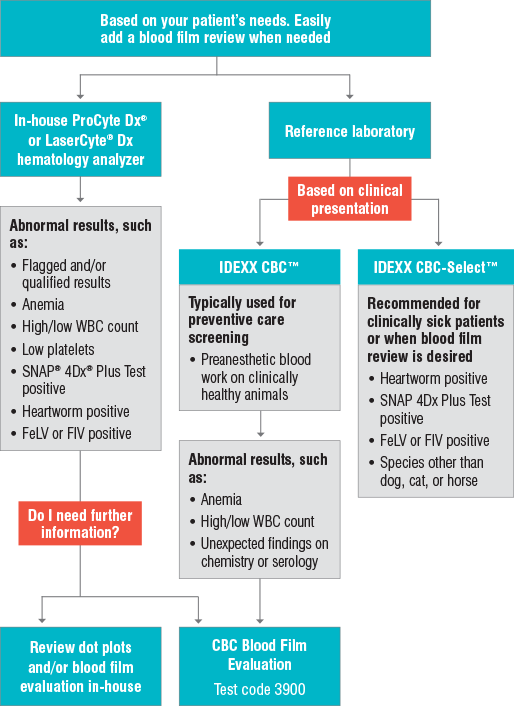
IDEXX CBC typically used for preventive care screening
The IDEXX CBC is the appropriate and most cost-effective option for routine preanesthetic or preventive care screenings on clinically healthy animals. Available for canine, feline, and equine patients, this option provides an automated CBC, including hemogram, five-part differential, platelets, platelet indices, as well as (for canine and feline patients) reticulocyte count and reticulocyte hemoglobin.
IDEXX CBC-Select recommended for sick patient diagnostics
The IDEXX CBC-Select is the option recommended for sick patients. It includes the same components as the IDEXX CBC, as well as a blood film evaluation by a technician to evaluate for abnormal red and white blood cell morphology, blood parasites, unclassified cells, and immature cell lines. A clinical pathologist review is automatically included when results are markedly abnormal or when unclassified cells are seen based on specific established criteria.
CBC options from IDEXX Reference Laboratories
When viewing IDEXX test results in VetConnect PLUS, access differential diagnoses (including those for reticulocyte and reticulocyte hemoglobin) instantly, helping you interpret and act on any results that are abnormal for your patient.
Options for abnormal CBC results
The IDEXX CBC is generally an excellent and economical choice when performing preventive care screening on a clinically normal animal. Sometimes, however, a patient who appears outwardly healthy may in fact have abnormal CBC or chemistry results. A positive heartworm antigen or tick/vector screen may also be an indication to evaluate a blood film review in a clinically healthy animal.
IDEXX Reference Laboratories offers the CBC Blood Film Evaluation. This add-on (test code 3900) allows you to request an evaluation of the blood film by a technician on your previously submitted IDEXX CBC. See order options for more information.
Choose the right CBC for your patient
Ordering options
| Test code | Test name and contents |
| 375 | IDEXX CBC |
| 300 | IDEXX CBC-Select |
| 3900 | CBC Blood Film Evaluation Blood film evaluation performed by a technician for RBC and WBC morphology and parasite screen. Pathologist review automatically performed when results are markedly abnormal based on established guidelines or if unclassified cells are seen. Note: Add on to an IDEXX CBC previously performed at IDEXX Reference Laboratories or on an in-house IDEXX hematology analyzer (ProCyte Dx or LaserCyte Dx). Must be ordered within 72 hours of initial CBC. |
| 399 | Pathologist Review of CBC Add on to an IDEXX CBC, avian/exotic CBC, or IDEXX CBC-Select. For IDEXX CBC-Select requests, a pathologist review is included and will be automatically performed at no additional charge if specific established criteria are met (see protocol information in the Online Test Directory at vetconnectplus.com). Test code 399 is not required for the automatic reflexed pathologist review on IDEXX CBC-Select requests. |
Specimen requirements: 1 mL LTT
Turnaround time: 8:00 a.m.; allow 1–2 working days for pathologist review of CBC if indicated.
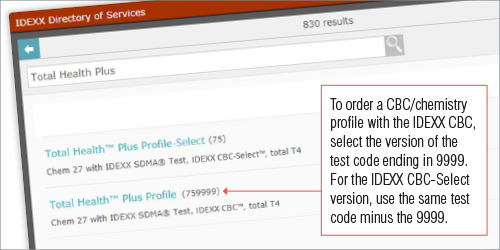
To order a profile with your desired CBC options
Customer support services
IDEXX supports your practice with our customer support, technical support, and medical consulting services teams, including our diagnostic support veterinarians and board-certified veterinary specialists.
*If automated results fall outside of established guidelines and the specimen is considered significantly abnormal, a blood film evaluation by a technician is performed.
†The criteria for a pathologist review were established by an international team of pathologists, internal medicine specialists, and operations specialists and have been endorsed by the IDEXX Medical Review Board.